Abstract
Leukemia stem cells (LSCs) represent a small portion (<0.1%) of chemoresistant cells that persist after initial treatment and are primarily responsible for relapse in acute myeloid leukemia (AML) patients. Over time LSCs repopulate the disease due to their ability to self-renew and produce differentiated progeny. Many studies have confirmed that a higher frequency of LSCs at diagnosis correlates with a significant decrease in relapse-free survival. Since LSCs are resistant to currently used chemotherapies, new therapies are needed that disrupt their repopulation capacity, thus eliminating the source of relapse and leading to long-term cures.
AML cells reside predominantly in the bone marrow (BM) which is characterized by intrinsic hypoxic conditions. Cellular adaptations to hypoxia can also confer a survival advantage by providing a means of resisting chemotherapy. One such mechanism is autophagy, a pro-survival recycling process used to reallocate nutrients and remove damaged organelles. Our lab has previously shown human AML cell lines cultured under hypoxia (1% O2) upregulate autophagy. Additionally, pharmacological inhibition of autophagy induces AML cell death, an effect which was significantly enhanced under hypoxia vs normoxia (21% O2). This suggests a reliance of AML cells on autophagy for survival under hypoxic conditions reflecting the natural BM microenvironment.
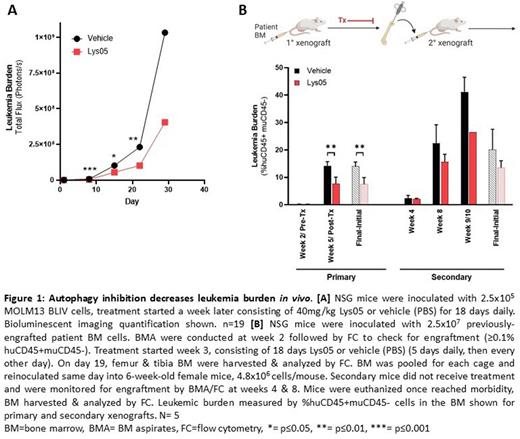
The clinical development of autophagy inhibitors has been limited due to lack of in vivo efficacy. Lys05 is a promising chloroquine derivative with increased potency and therapeutic potential. To examine the efficacy of autophagy inhibition in vivo, NSG mice systemically engrafted with luciferase-expressing human AML cell line MOLM13-BLIV were treated with Lys05 for 18 days. Lys05 significantly reduced leukemia burden and led to a significant increase in overall survival. These data demonstrate the importance of autophagy in AML survival in vivo and that an efficacious concentration of Lys05 can be achieved in vivo without major toxicities.
As LSCs are the critical target for novel therapeutics, we examined the impact of autophagy inhibition on LSCs. We performed colony formation unit (CFU) assays using multiple AML patient BM samples representing a variety of mutational profiles. Samples were treated with Lys05 and cultured under normoxic or hypoxic conditions. Lys05 significantly decreased the number of colonies under both oxygen conditions, with a further decrease in colony number under hypoxia. This suggests that autophagy may be crucial for LSC survival in the hypoxic BM and supports autophagy inhibition as a potential therapeutic method of eliminating LSCs.
To functionally measure the effect of autophagy inhibition on the repopulation capacity of LSCs in vivo, we utilized serially transplanted patient-derived xenografts (PDXs). Serial transplants are the standard for functionally defining stem cells in vivo as it tests the ability to self-renew and repopulate disease. AML PDX mice established by our lab were treated with Lys05 following engraftment confirmation (≥0.1% huCD45+muCD45- in BM). Lys05 significantly decreased leukemia burden in 1° xenografts, confirming our findings in our MOLM13-BLIV model. Additionally, autophagy inhibition caused a decrease in the LSC population (%CD34+CD38-), supporting the results from our in vitro CFU assays. Post-treatment, BM was harvested and transplanted into new mice. These 2° mice did not receive treatment and were monitored via BM aspiration every 2-4 weeks for engraftment. Excitingly, 2° xenografts that received Lys05 treatment during the 1° xenograft had a decrease in both leukemia burden and LSCs vs those that received vehicle during the 1° xenograft. These results suggest that autophagy inhibition has lasting effects on AML cells and indicates that autophagy inhibition interferes with the ability of LSCs to repopulate disease.
Here we have demonstrated that autophagy inhibitor Lys05 is efficacious in the hypoxic BM environment, decreasing leukemia burden and prolonging survival. We have also shown that autophagy inhibition impacts AML LSCs, reducing their ability to repopulate disease upon transplantation. These results suggest autophagy plays a key role in the maintenance of AML LSCs and suggests that inhibition of autophagy by pharmacological agents such as Lys05 may be a viable therapeutic method of eliminating LSCs.
Disclosures
Wang:Dava Oncology: Consultancy, Speakers Bureau; Rafael Pharmaceuticals: Other: Data Safety Monitoring Committee; Gilead: Consultancy, Honoraria, Other: Advisory Board; Jazz Pharmaceuticals: Consultancy, Honoraria, Other: Advisory Board; Mana Therapeutics: Consultancy, Honoraria; GlaxoSmithKline: Consultancy, Honoraria, Other: Advisory Board; Abbvie: Consultancy, Honoraria, Other: member of data monitoring committee ; Novartis: Consultancy, Honoraria, Other: Advisory Board; PTC Therapeutics: Consultancy, Honoraria, Other: Advisory Board; Kite Pharmaceuticals: Consultancy, Honoraria, Other: Advisory Board; Kura Oncology: Consultancy, Honoraria, Other: Advisory Board, Steering Committee, Speakers Bureau; Stemline Therapeutics: Consultancy, Honoraria, Other: Advisory Board, Speakers Bureau; Astellas: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria, Other: Advisory Board, Speakers Bureau; BMS/Celgene: Membership on an entity's Board of Directors or advisory committees; Genentech: Consultancy; Takeda: Consultancy, Honoraria, Other: Advisory Board; Daiichi Sankyo: Consultancy, Honoraria, Other: Advisory Board; Macrogenics: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.